 
Fully synthetic human combinatorial antibody libraries (HuCAL) based on modular consensus frameworks and CDRs randomized with trinucleotides. A large non-immunized human Fab fragment phage library that permits rapid isolation and kinetic analysis of high affinity antibodies. Human antibodies with sub-nanomolar affinities isolated from a large non-immunized phage display library. Isolation of high affinity human antibodies directly from large synthetic repertoires. Overview of antibody phage-display technology and its applications. Making antibody by phage display technology. Winter, G., Griffiths, A.D., Hawkins, R.E. A large array of human monoclonal antibodies to type 1 human immunodeficiency virus from combinatorial libraries of asymptomatic individuals. By-passing immunization: human antibodies from V-gene libraries displayed on phage. Making antibody fragments using phage display libraries. 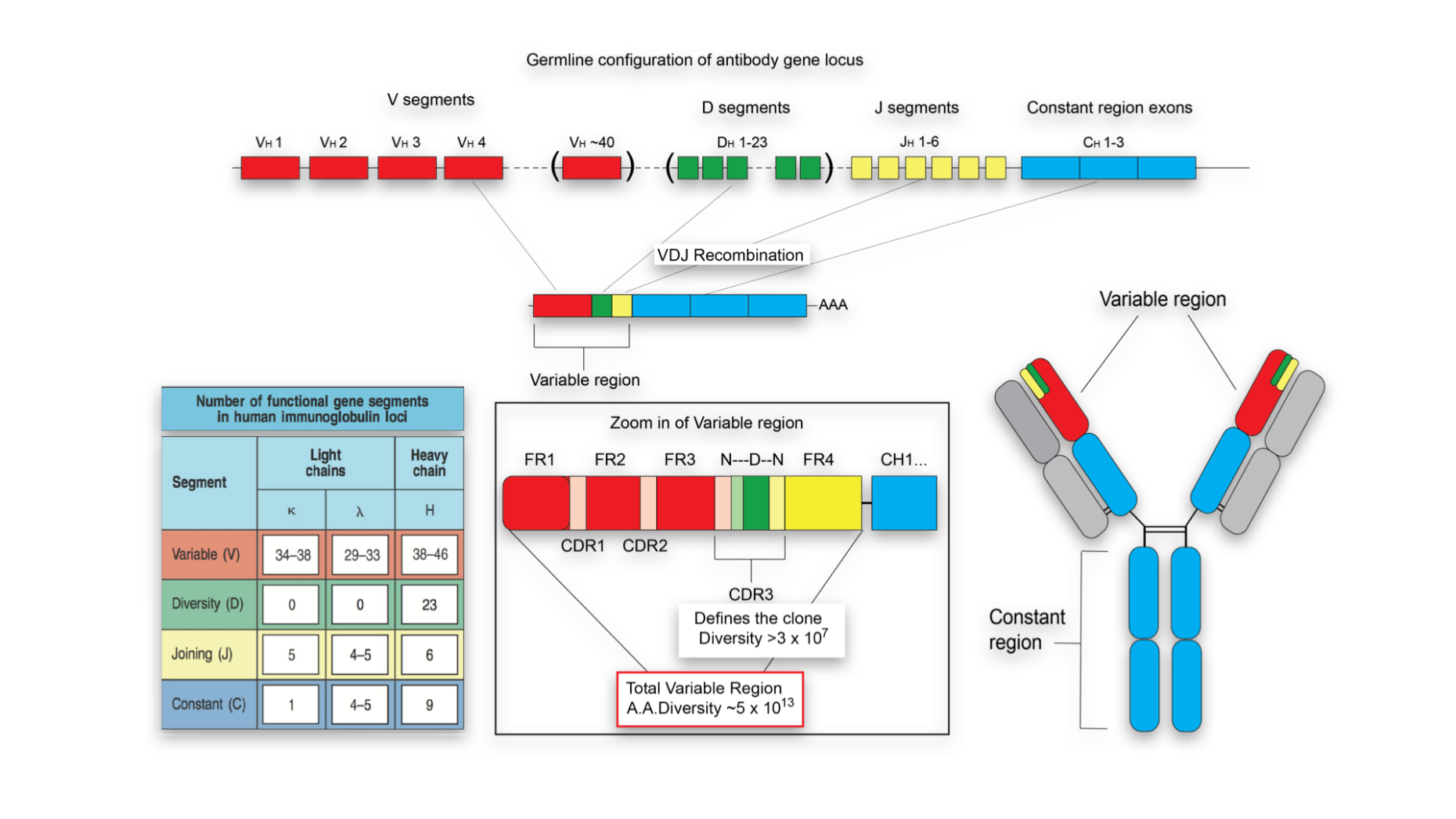
Phage antibodies: filamentous phage displaying antibody variable domains. McCafferty, J., Griffiths, A.D., Winter, G. 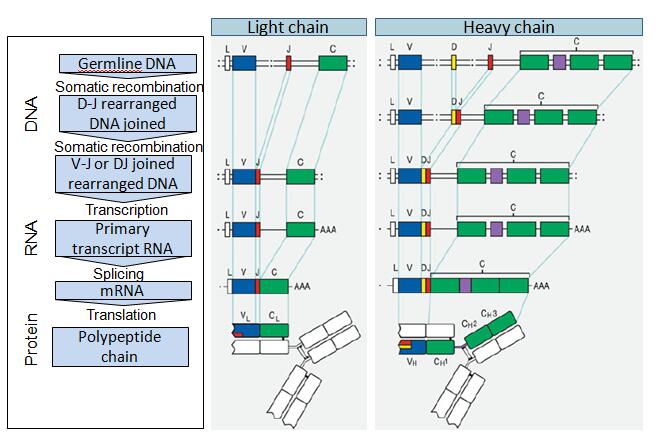
Generation of a large combinatorial library of the immunoglobulin repertoire in phage lambda. Binding activities of a repertoire of single immunoglobulin variable domains secreted from Escherichia coli. Ward, E.S., Gussow, D., Griffiths, A.D., Jones, P.T. Cloning immunoglobulin variable domains for expression by the polymerase chain reaction. With the first antibody of this new generation, adalimumab (Humira, a human IgG1 specific for human tumor necrosis factor (TNF)), already approved for therapy and with many more in clinical trials, these recombinant antibody technologies will provide a solid basis for the discovery of antibody-based biopharmaceuticals, diagnostics and research reagents for decades to come. Automation of the technology is making it possible to identify hundreds of different antibody leads to a single therapeutic target. Innovative strategies enable directed evolution of binding sites with ultra-high affinity, high stability and increased potency, sometimes to a level that cannot be achieved by immunization. Clever antibody library designs and selection concepts are now able to identify mAb leads with virtually any specificity. These technologies are now widely exploited to build human antibodies with high affinity and specificity. During the past decade several display methods and other library screening techniques have been developed for isolating monoclonal antibodies (mAbs) from large collections of recombinant antibody fragments. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
